 | |
| Names | |
|---|---|
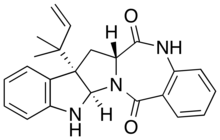
| IUPAC name (2R,10S,12R)-10-(2-methylbut-3-en-2-yl)-1,3,14-triazapentacyclohenicosa-4,6,8,15,17,19-hexaene-13,21-dione | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C23H23N3O2 |
| Molar mass | 373.456 g·mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa). Infobox references | |
Aszonalenin is an alkaloid which is produced by Neosartorya and Aspergillus species. Aszonalenin is a neurotoxin.
References
- "Aszonalenin". PubChem.
- Whittall, John; Sutton, Peter W. (16 April 2012). Practical Methods for Biocatalysis and Biotransformations 2. John Wiley & Sons. p. 254. ISBN 978-1-119-94341-9.
- Grundon, M. F. (1983). Alkaloids. Royal Society of Chemistry. p. 214. ISBN 978-0-85186-367-2.
- Lubertozzi, David M. (2009). "Developing Aspergillus as a Host for Heterologous Expression". Biotechnology Advances. 27 (1). University of California, Berkeley: 53–75. doi:10.1016/j.biotechadv.2008.09.001. PMID 18840517.
Further reading
- Ruchti, Jonathan; Carreira, Erick M. (2014). "Ir-Catalyzed Reverse Prenylation of 3-Substituted Indoles: Total Synthesis of (+)-Aszonalenin and (–)-Brevicompanine B". J. Am. Chem. Soc. 136 (48): 16756–16759. doi:10.1021/ja509893s. PMID 25365411.
- Kimura, Yasuo; Hamasaki, Takashi; Nakajima, Hiromitsu; Isogai, Akira (1982). "Structure of aszonalenin, a new metabolite of Aspergillus zonatus". Tetrahedron Letters. 23 (2): 225–228. doi:10.1016/S0040-4039(00)86791-X. ISSN 0040-4039.
This article about an alkaloid is a stub. You can help Misplaced Pages by expanding it. |